|
2/9/2024 0 Comments Electron capture decay 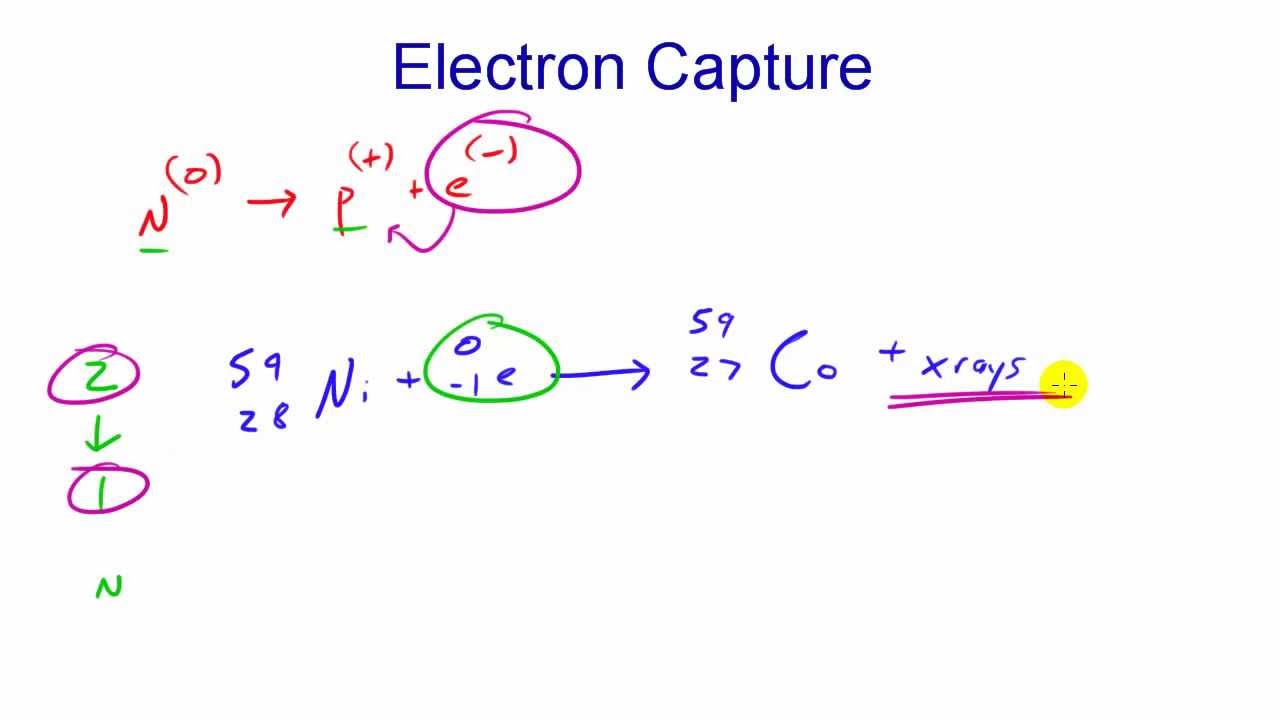
In the case of the β+ decay, each decaying nucleus emits a positron and a neutrino, reducing its atomic number by one while the mass number stays the same.Ī positron does not exist for a long period of time in the presence of matter. Similar to an antineutrino, a neutrino has no electric charge nor rest mass. If the number of neutrons in a nucleus is smaller than the number of protons in an unstable nucleus, a proton will undergo the following transformation: p -> n + β+ + ν e, i.e. a proton will be converted into a neutron with the emission of a positron (β+ or beta plus particle) and a neutrino. The example of importance in radiology is the decay of cobalt-60: 60Co -> 60Ni + β- + ν*.Īnother example is iodine-131 which undergoes beta minus decay into xenon-131 7 by increasing atomic number by 1 while keeping the same mass number 8. There are numerous examples of beta minus emitters in nature like 14C, 40K, 3H, 60Co etc. The mass number remains the same but the atomic number increases by one. The antineutrino has no rest mass nor electric charge and does not interact readily with the matter.įor the isotopes that undergo β-decay, each nucleus emits an electron and an antineutrino. If the number of neutrons in a nucleus is in excess, a neutron will undergo the following transformation: n -> p + β- + ν e*, i.e., a neutron will be converted into a proton with the emission of a beta-minus particle ( electron) and an antineutrino.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
